Strategy & CMC advisory
Expertise in pharmaceutical strategy and product development
from development to industrialization
and across the whole commercial life cycle
Technical Life Cycle Management
Seamless API launch: from concept to market with optimal return on investment
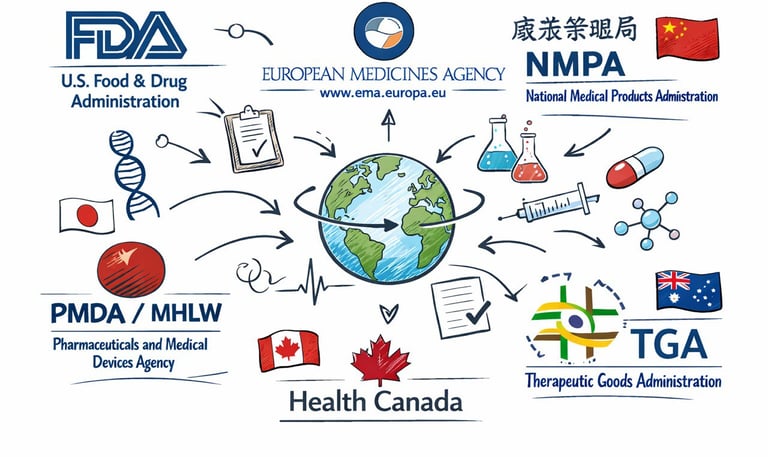
Regulatory Perspective
Advice on regulatory strategy for worldwide submissions, ensuring compliance and smooth approval processes.


Risk Management
Support for appropriate quality assurance processes, specification setting and risk assessments to enable short product development timelines and excellent OTIF (on time in full).
Development & Industrialization Perspective
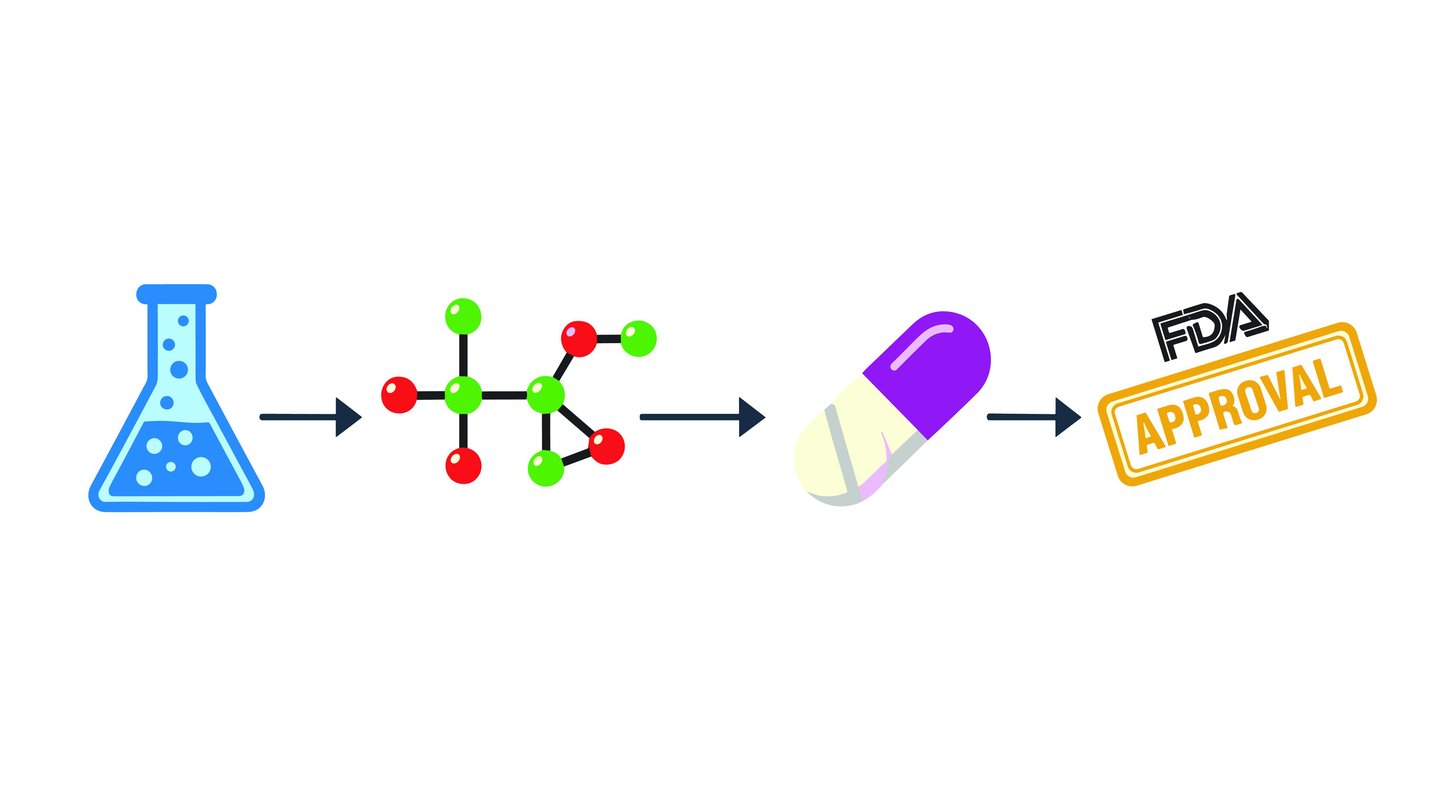
Help to pick the best synthetic routes, production and supply concepts including Make or Buy and Cost of Goods assessments. Coaching for effective project and milestone planning.
Contact
info@cfw.limited
+3579 5949 399
© 2026. All rights reserved.
PHONE
Legal Notice
Website owner & responsible for content:
Frank Wetterich on behalf of
CFW – Consultancy Frank Wetterich Ltd.
Legal Form: Limited Company (Ltd)
Registration Number: HE 484564
Tax Identification Number: TIN 60274450B
